Osmium(III) chloride
| |
| Names | |
|---|---|
| Other names
trichloroosmium, osmium trichloride
| |
| Identifiers | |
| |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.033.247 |
| EC Number |
|
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| Cl3Os | |
| Molar mass | 296.58 g·mol−1 |
| Appearance | black-brown crystals |
| Melting point | 560 °C (1,040 °F; 833 K) |
| soluble | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
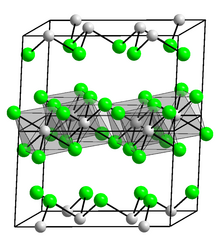
Osmium(III) chloride is an inorganic chemical compound of osmium metal and chlorine with the chemical formula OsCl3.[1][2]
Synthesis[edit]
Osmium(III) chloride can be made by a reaction of chlorine with osmium:
- 2Os + 3Cl2 → 2OsCl3
It can also be made by heating of osmium(IV) chloride:
- 2OsCl4 → 2OsCl3 + Cl2
Physical properties[edit]

Osmium(III) chloride forms black-brown crystals.[3]
Osmium(III) chloride forms a hydrate of the composition OsCl3·3H2O with dark green crystals.[4]
Uses and reactions[edit]
Osmium(III) chloride hydrate is used as a precursor material for the production of dichlorodihydridoosmium complex compounds and other compounds.[5]
It is the precursor to a variety of arene complexes.[6]
References[edit]
- ^ "Osmium(III) chloride". Sigma Aldrich. Retrieved 31 March 2023.
- ^ Lide, David R. (19 June 2003). 1998 Freshman Achievement Award. CRC Press. p. 4-79. ISBN 978-0-8493-0594-8. Retrieved 31 March 2023.
- ^ "Osmium(III) chloride". heraeus.com. Retrieved 31 March 2023.
- ^ "Osmium(III) chloride hydrate, Thermo Scientific Chemicals | Fisher Scientific". fishersci.se. Retrieved 31 March 2023.
- ^ "Osmium(III) chloride hydrate". Sigma Aldrich. Retrieved 31 March 2023.
- ^ Bell, Andrew G.; Koźmiński, Wiktor; Linden, Anthony; von Philipsborn, Wolfgang (1996). "187Os NMR Study of (η6-Arene)osmium(II) Complexes: Separation of Electronic and Steric Ligand Effects". Organometallics. 15 (14): 3124–3135. doi:10.1021/om960053i.
