Beryllium nitride
| |
| Names | |
|---|---|
| IUPAC name
Beryllium nitride
| |
| Other names
triberyllium dinitride
| |
| Identifiers | |
3D model (JSmol)
|
|
| ECHA InfoCard | 100.013.757 |
| EC Number |
|
| UNII | |
| |
| |
| Properties | |
| Be3N2 | |
| Molar mass | 55.051 g·mol−1 |
| Appearance | yellow or white powder |
| Density | 2.71 g/cm3 |
| Melting point | 2,200 °C (3,990 °F; 2,470 K) |
| Boiling point | 2,240 °C (4,060 °F; 2,510 K) (decomposes) |
| decomposes | |
| Solubility | decomposes in solutions of acid and base |
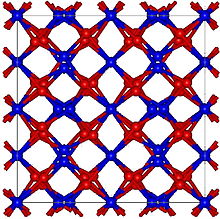
| Structure | |
| Cubic, cI80, SpaceGroup = Ia-3, No. 206 (α form) | |
| Hazards | |
| NIOSH (US health exposure limits): | |
PEL (Permissible)
|
TWA 0.002 mg/m3 C 0.005 mg/m3 (30 minutes), with a maximum peak of 0.025 mg/m3 (as Be)[1] |
REL (Recommended)
|
Ca C 0.0005 mg/m3 (as Be)[1] |
IDLH (Immediate danger)
|
Ca [4 mg/m3 (as Be)][1] |
| Related compounds | |
Other cations
|
Calcium nitride Magnesium nitride |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Beryllium nitride, Be3N2, is a nitride of beryllium. It can be prepared from the elements at high temperature (1100–1500 °C);[2] unlike beryllium azide or BeN6, it decomposes in vacuum into beryllium and nitrogen.[2] It is readily hydrolysed forming beryllium hydroxide and ammonia.[2] It has two polymorphic forms cubic α-Be3N2 with a defect anti-fluorite structure, and hexagonal β-Be3N2.[2] It reacts with silicon nitride, Si3N4 in a stream of ammonia at 1800–1900 °C to form BeSiN2.[2]
Preparation[edit]
Beryllium nitride is prepared by heating beryllium metal powder with dry nitrogen in an oxygen-free atmosphere in temperatures between 700 and 1400 °C.
- 3Be + N2 → Be3N2
Uses[edit]
It is used in refractory ceramics[3] as well as in nuclear reactors.
It is used to produce radioactive carbon-14 for tracer applications by the 14
7N + n → 14
6C + p reaction. It is favoured due to its stability, high nitrogen content (50%), and the very low cross section of beryllium for neutrons.[4]
Reactions[edit]
Beryllium nitride reacts with mineral acids producing ammonia and the corresponding salts of the acids:
- Be3N2 + 6 HCl → 3 BeCl2 + 2 NH3
In strong alkali solutions, a beryllate forms, with evolution of ammonia:
- Be3N2 + 6 NaOH → 3 Na2BeO2 + 2 NH3
Both the acid and alkali reactions are brisk and vigorous. Reaction with water, however, is very slow:
- Be3N2 + 6 H2O → 3 Be(OH)2 + 2 NH3
Reactions with oxidizing agents are likely to be violent. It is oxidized when heated at 600 °C in air.
References[edit]
- ^ a b c NIOSH Pocket Guide to Chemical Hazards. "#0054". National Institute for Occupational Safety and Health (NIOSH).
- ^ a b c d e Egon Wiberg, Arnold Frederick Holleman (2001) Inorganic Chemistry, Elsevier ISBN 0-12-352651-5
- ^ Hugh O. Pierson, 1996, Handbook of Refractory Carbides and Nitrides: Properties, Characteristics, Processing, and Applications, William Andrew Inc.,ISBN 0-8155-1392-5
- ^ Shields, R. P. (1956-02-01). THE PRODUCTION OF C$sup 14$ BY THE Be$sub 3$N$sub 2$ PROCESS (Report). Oak Ridge National Lab. (ORNL), Oak Ridge, TN (United States).
