Transition-metal allyl complex
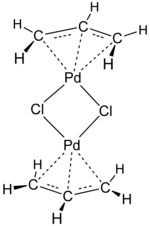
Transition-metal allyl complexes are coordination complexes with allyl and its derivatives as ligands. Allyl is the radical with the connectivity CH2CHCH2, although as a ligand it is usually viewed as an allyl anion CH2=CH−CH2−, which is usually described as two equivalent resonance structures.
Examples
[edit]The allyl ligand is commonly in organometallic chemistry. Usually, allyl ligands bind to metals via all three carbon atoms, the η3-binding mode. The η3-allyl group is classified as an LX-type ligand in the Green LXZ ligand classification scheme, serving as a 3e– donor using neutral electron counting and 4e– donor using ionic electron counting.
Scope
[edit]Commonly, allyl ligands occur in mixed ligand complexes. Examples include (η3-allyl)Mn(CO)4 and CpPd(allyl).
Substituents on the allyl group are also common, e.g. 2-methallyl.[1]
Homoleptic complexes
[edit]- bis(allyl)nickel[2]
- bis(allyl)palladium[2]
- bis(allyl)platinum[2]
- tris(allyl)chromium[2]
- tris(allyl)rhodium[3]
- tris(allyl)iridium[3]
Chelating bis(allyl) complexes
[edit]
1,3-Dienes such as butadiene and isoprene dimerize in the coordination spheres of some metals, giving chelating bis(allyl) complexes. Such complexes also arise from ring-opening of divinylcyclobutane. Chelating bis(allyl) complexes are intermediates in the metal-catalyzed dimerization of butadiene to give vinylcyclohexene and cycloocta-1,5-diene.[4]
Allyl σ ligands
[edit]Complexes with η1-allyl ligands (classified as X-type ligands) are also known. One example is CpFe(CO)2(η1-C3H5), in which only the methylene group is attached to the Fe centre (i.e., it has the connectivity [Fe]–CH2–CH=CH2). As is the case for many other η1-allyl complexes, the monohapticity of the allyl ligand in this species is enforced by the 18-electron rule, since CpFe(CO)2(η1-C3H5) is already an 18-electron complex, while an η3-allyl ligand would result in an electron count of 20 and violate the 18-electron rule. Such complexes can convert to the η3-allyl derivatives by dissociation of a neutral (two-electron) ligand L. For CpFe(CO)2(η1-C3H5), dissociation of L = CO occurs under photochemical conditions:[5]
- CpFe(CO)2(η1-C3H5) → CpFe(CO)(η3-C3H5) + CO
Synthetic methods
[edit]Allyl complexes are often generated by oxidative addition of allylic halides to low-valent metal complexes. This route is used to prepare (allyl)2Ni2Cl2:[1][6]
- 2 Ni(CO)4 + 2 ClCH2CH=CH2 → Ni2(μ-Cl)2(η3-C3H5)2 + 8 CO
A similar oxidative addition involves the reaction of allyl bromide to diiron nonacarbonyl.[7] The oxidative addition route has also been used to prepared Mo(II) allyl complexes:[8]
- Mo(CO)3(pyridine)3 + BrCH2CH=CH2 → Mo(CO)2(Br)(C3H5)(pyridine)2 + pyridine + CO
Other methods of synthesis involve addition of nucleophiles to η4-diene complexes and hydride abstraction from alkene complexes.[3] For example, palladium(II) chloride attacks alkenes to give first an alkene complex, but then abstracts hydrogen to give a dichlorohydridopalladium alkene complex, and then eliminates hydrogen chloride:[9]
- PdCl2 + >C=CHCH< → Cl2Pd–(η2-(>CCHCH<)) → Cl2Pd(H)⚟(>CCHC<) → ClPd⚟(>CCHC<) + HCl
One allyl complex can transfer an allyl ligand to another complex.[10] An anionic metal complex can displace a halide, to give an allyl complex. However, if the metal center is coordinated to 6 or more other ligands, the allyl may end up "trapped" as a σ (η1-) ligand. In such circumstances, heating or irradiation can dislocate another ligand to free up space for the alkene-metal bond.[11]
In principle, salt metathesis reactions can adjoin an allyl ligand from an allylmagnesium bromide or related allyl lithium reagent.[3] However, the carbanion salt precursors require careful synthesis, as allyl halides readily undergo Wurtz coupling. Mercury and tin allyl halides appear to avoid this side-reaction.[12]
Benzyl complexes
[edit]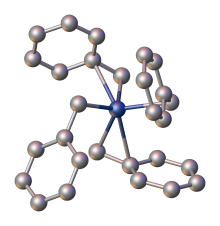
Benzyl and allyl ligands often exhibit similar chemical properties. Benzyl ligands commonly adopt either η1 or η3 bonding modes. The interconversion reactions parallel those of η1- or η3-allyl ligands:
- CpFe(CO)2(η1-CH2Ph) → CpFe(CO)(η3-CH2Ph) + CO
In all bonding modes, the benzylic carbon atom is more strongly attached to the metal as indicated by M-C bond distances, which differ by ca. 0.2 Å in η3-bonded complexes.[14] X-ray crystallography demonstrate that the benzyl ligands in tetrabenzylzirconium are highly flexible. One polymorph features four η2-benzyl ligands, whereas another polymorph has two η1- and two η2-benzyl ligands.[13]
Applications
[edit]Allyl complexes are often discussed in academic research,[15][16][17][18] but few have commercial applications. A popular allyl complex is allyl palladium chloride.[19]
The reactivity of allyl ligands depends on the overall complex, although the influence of the metal center can be roughly summarized as[20]
- (more reactive) Fe ≫ Pd > Mo > W (less reactive)
Such complexes are usually electrophilic (i.e., react with nucleophiles), but nickel allyl complexes are usually nucleophilic (resp. with electrophiles).[21] In the former case, the addition may occur at unusual locations, and can be useful in organic synthesis.[22]
References
[edit]- ^ a b Semmelhack, Martin F.; Helquist, Paul M. (1972). "Reactions of Aryl Halides with π-Allylnickel Halides: Methallylbenzene". Organic Syntheses. 52: 115. doi:10.15227/orgsyn.052.0115.
- ^ a b c d O'Brien, S.; Fishwick, M.; McDermott, B.; Wallbridge, M. G. H.; Wright, G. A. (1972). "Isoleptic Allyl Derivatives of Various Metals". Inorganic Syntheses. Vol. 13. pp. 73–79. doi:10.1002/9780470132449.ch14. ISBN 978-0-470-13244-9.
- ^ a b c d Kevin D. John; Judith L. Eglin; Kenneth V. Salazar; R. Thomas Baker; Alfred P. Sattelberger (2014). "Tris(Allyl)Iridium and -Rhodium". Inorganic Syntheses: Volume 36. Vol. 36. p. 165. doi:10.1002/9781118744994.ch32. ISBN 978-1-118-74499-4.
- ^ Hirano, Masafumi; Sakate, Yumiko; Komine, Nobuyuki; Komiya, Sanshiro; Wang, Xian-qi; Bennett, Martin A. (2011). "Stoichiometric Regio- and Stereoselective Oxidative Coupling Reactions of Conjugated Dienes with Ruthenium(0). A Mechanistic Insight into the Origin of Selectivity". Organometallics. 30 (4): 768–777. doi:10.1021/om100956f.
- ^ Fish, R. W.; Giering, W. P.; Marten, D.; Rosenblum, M. (1976-01-27). "Thermal and photochemical interconversions of isomeric monocarbonyl η5-Cyclopentadienyl(η3-allyl)iron complexes". Journal of Organometallic Chemistry. 105 (1): 101–118. doi:10.1016/S0022-328X(00)91977-6. ISSN 0022-328X.
- ^ Craig R. Smith; Aibin Zhang; Daniel J. Mans; T. V. RajanBabu (2008). "(R)-3-methyl-3-phenyl-1-pentene Via Catalytic Asymmetric Hydrovinylation". Org. Synth. 85: 248–266. doi:10.15227/orgsyn.085.0248. PMC 2723857. PMID 19672483.
- ^ Putnik, Charles F.; Welter, James J.; Stucky, Galen D.; d'Aniello, M. J.; Sosinsky, B. A.; Kirner, J. F.; Muetterties, E. L. (1978). "Metal clusters in catalysis. 15. A Structural and Chemical Study of a Dinuclear Metal Complex, Hexacarbonylbis(η3-2-propenyl)diiron(Fe-Fe)". Journal of the American Chemical Society. 100 (13): 4107–4116. doi:10.1021/ja00481a020.
- ^ Pearson, Anthony J.; Schoffers, Elke (1997). "Tricarbonyltris(pyridine)molybdenum: A Convenient Reagent for the Preparation of (π-Allyl)molybdenum Complexes". Organometallics. 16 (24): 5365–5367. doi:10.1021/om970473n.
- ^ Pearson, A. J. (1987). "Transition metal-stabilized carbocations in organic synthesis". In Hartley, Frank R. (ed.). The Chemistry of the Metal–Carbon Bond. Vol. 4. John Wiley & Sons. p. 911. doi:10.1002/9780470771778. ISBN 978-0-470-77177-8.
- ^ Powell 1982, p. 328.
- ^ Powell 1982, p. 329.
- ^ Powell, P. (1982). "Synthesis of η3-allyl complexes". In Hartley, Frank R.; Patai, Saul (eds.). The Chemistry of the Metal–Carbon Bond. Vol. 1: The structure, preparation, thermochemistry and characterization of organometallic compounds. Chichester, UK: Interscience (published April 1987). pp. 326–8. ISBN 0471100587.
- ^ a b Rong, Yi; Al-Harbi, Ahmed; Parkin, Gerard (2012). "Highly Variable Zr–CH2–Ph Bond Angles in Tetrabenzylzirconium: Analysis of Benzyl Ligand Coordination Modes". Organometallics. 31pages=8208–8217 (23): 8208–8217. doi:10.1021/om300820b.
- ^ Trost, Barry M.; Czabaniuk, Lara C. (2014). "Structure and Reactivity of Late Transition Metal η3-Benzyl Complexes". Angew. Chem. Int. Ed. 53 (11): 2826–2851. doi:10.1002/anie.201305972. PMID 24554487.
- ^ Consiglio, Giambattista; Waymouth, Robert M. (1989). "Enantioselective homogeneous catalysis involving transition-metal-allyl intermediates". Chemical Reviews. 89: 257–276. doi:10.1021/cr00091a007.
- ^ Li, Chien-Le; Liu, Rai-Shung (2000). "Synthesis of Heterocyclic and Carbocyclic Compounds via Alkynyl, Allyl, and Propargyl Organometallics of Cyclopentadienyl Iron, Molybdenum, and Tungsten Complexes". Chemical Reviews. 100 (8): 3127–3162. doi:10.1021/cr990283h. PMID 11749315.
- ^ Ley, Steven V.; Cox, Liam R.; Meek, Graham (1996). "(π-Allyl)tricarbonyliron Lactone Complexes in Organic Synthesis: A Useful and Conceptually Unusual Route to Lactones and Lactams". Chemical Reviews. 96: 423–442. doi:10.1021/cr950015t. PMID 11848759.
- ^ Welker, Mark E. (1992). "3 + 2 Cycloaddition reactions of transition-metal 2-alkynyl and .eta.1-allyl complexes and their utilization in five-membered-ring compound syntheses". Chemical Reviews. 92: 97–112. doi:10.1021/cr00009a004.
- ^ Tatsuno, Y.; Yoshida, T.; Otsuka, S. "(η3-allyl)palladium(II) Complexes" Inorganic Syntheses, 1990, volume 28, pages 342-345. ISBN 0-471-52619-3
- ^ Pearson 1987, pp. 932–933.
- ^ Chiusoli, G. P.; Salerno, G.; Tsuji J.; Sato F. (1985). "Carbon-carbon bond formation using η3-allyl complexes". In Hartley, Frank R.; Patai, Saul (eds.). The Chemistry of the Metal–Carbon Bond. Vol. 3: Carbon-carbon bond formation using organometallic compounds. Chichester, UK: John Wiley & Sons. pp. 143–205. ISBN 0471905577.
- ^ Hartwig, J. F. Organotransition Metal Chemistry, from Bonding to Catalysis; University Science Books: New York, 2010. ISBN 189138953X
